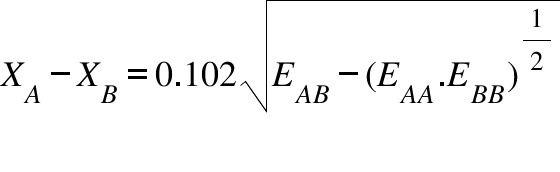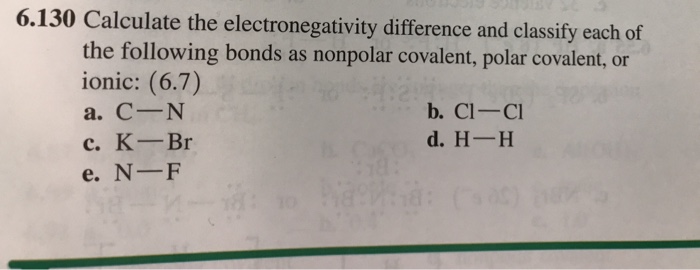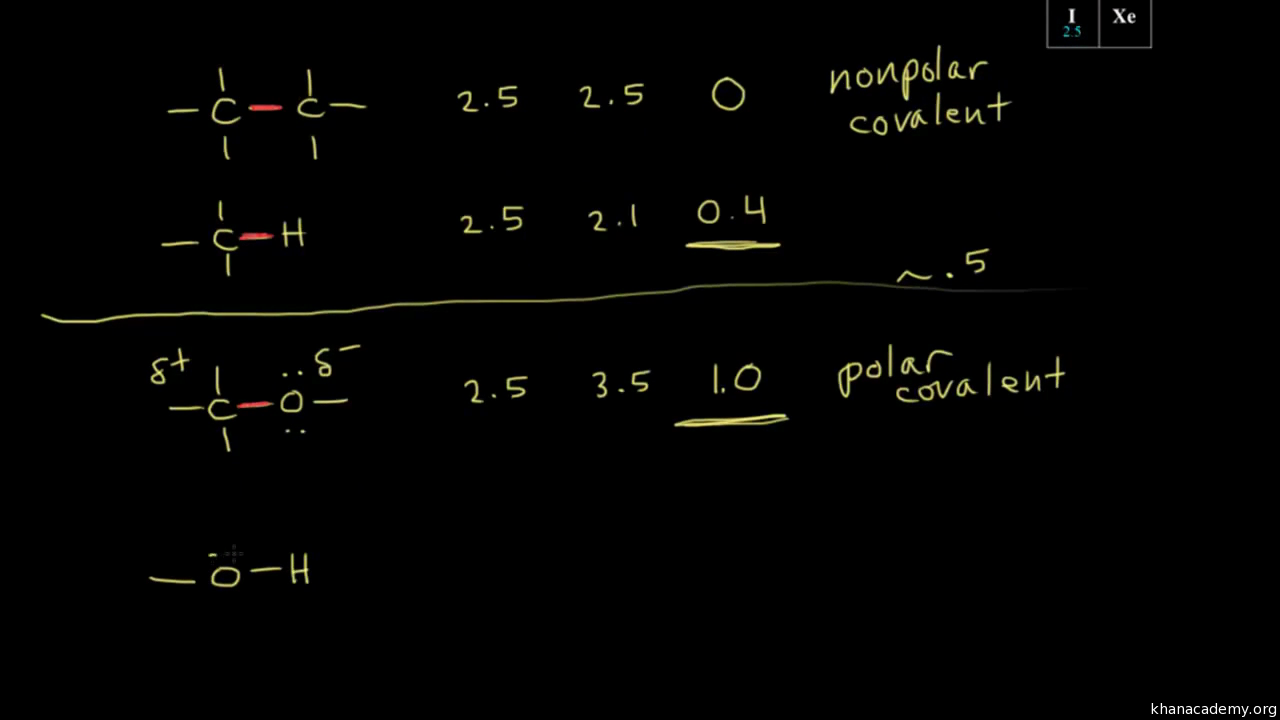Calculate electronegativity of carbon at Pauling scale Given that : ` E_(H-H) =104 .2 kcal "mol"^ - YouTube
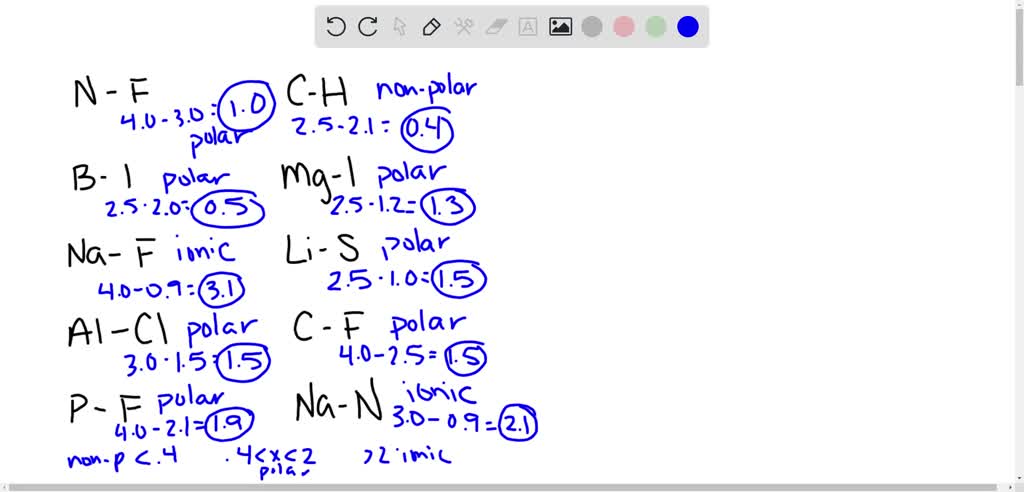
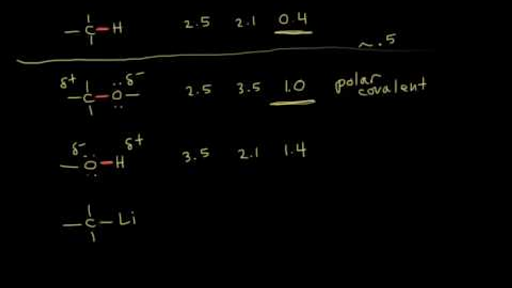
SOLVED: 1. Calculate the difference in electronegativity (ΔEN) for the bonds that would form between the groups of atoms listed below. State whether the bond would be ionic, polar covalent or non-polar

Question Video: Determining the Polarity of the Bond and Overall Molecule of Molecular Nitrogen | Nagwa

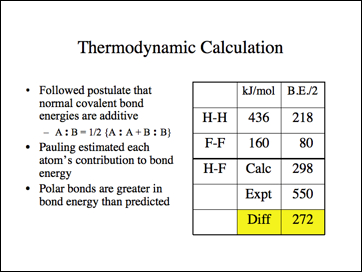
Calculate the electronegativity (10 time) of fluorine on Pauling scale form the following data: EH - H = 104.2kcal mol^-1 EF - F = 36.6kcal mol^-1 EH - F = 134.6kcal mol^-1 Given = XH = 2.1

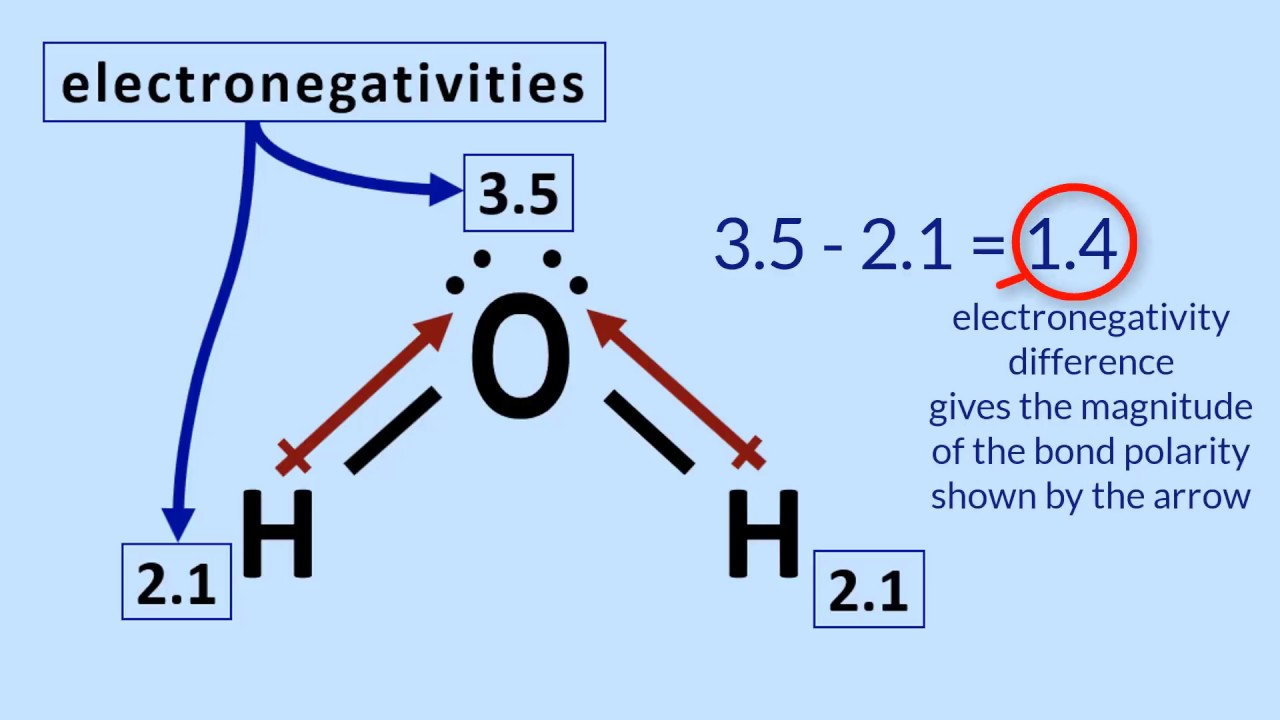
Electronegativity, Basic Introduction, Periodic Trends - Which Element Is More Electronegative? - YouTube